Informed Consent in the Genomic Medicine Era –
Current Practice, Normative Frameworks, Effective Solutions
Initiative Fact Sheet :: December 2020
Initiative Vision
A global framework for informed consent practice across genomic medicine – initially focused on gene therapy development and clinical translation – which is medically responsible, operationally sound, ethically resilient, and supported by a toolkit of open-source IC templates, content modules and assessment strategies.
Endpoints/Deliverables
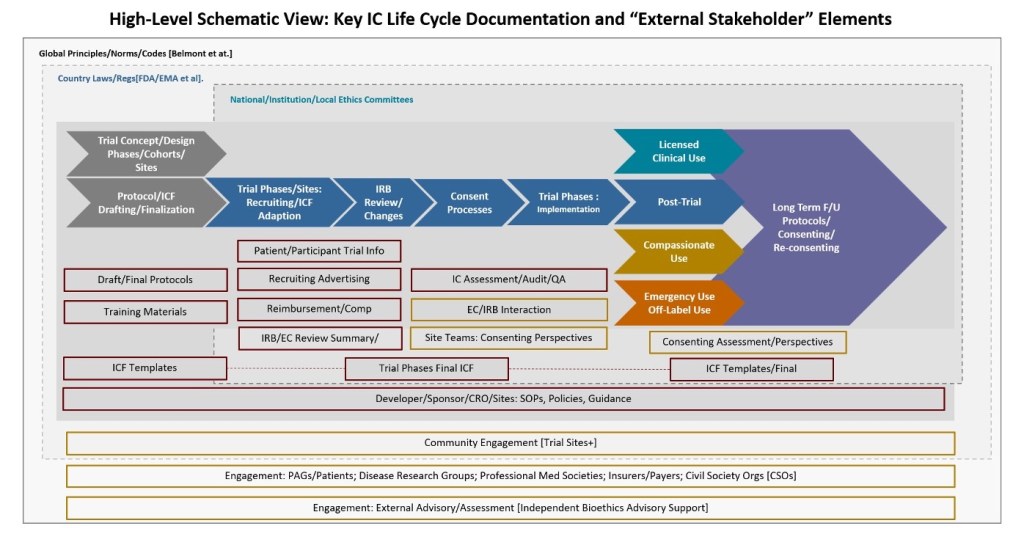
■ Systematic comparative assessment of IC practice across the gene therapy development life cycle including LTFU via a diverse cohort of programs indicative of disease areas under investigation/therapy modalities/geographies
▪ Active/ongoing engagement of patients/patient advocacy groups and other stakeholders in defining IC practice and metrics for gene therapy/genomic medicine
▪ Robust model of the full IC life cycle and key intervention/assessment points from trial protocol/ICF development to trial site/IRB-IEC refinement to direct consenting activity to clinical and LTFU contexts, etc.
▪ Toolkit of IC principles, templates, content building blocks and communications techniques, and assessment approaches [open source]
▪ Active piloting of principles/templates/toolkits in new trials, CU, clinical contexts, LTFU, enabling further assessment/refinement
Initiative Advisory Group [non-fiduciary; in formation – recruiting globally]
Dr. Anant Bhan
Adjunct Professor, Yenepoya (University), Mangaluru, India; Department of Health Research; Government of India – Gene Therapy Advisory and Evaluation Committee (GTAEC)
Barbara E Bierer, MD
Professor of Medicine, Harvard Medical School and Brigham and Women’s Hospital, Boston; Director of the Multi-Regional Clinical Trials Center (MRCT Center)
Jantina de Vries, DPhil
Associate Professor in Bioethics at the Department of Medicine of the University of Cape Town, South Africa; Co-PI of the H3Africa ELSI Collaborative Centre
Benjamin S. Wilfond, MD
Professor and Chief, Division of Bioethics; Professor, Pulmonary and Sleep Medicine, Department of Pediatrics; Adjunct Professor, Department of Bioethics and Humanities, University of Washington School of Medicine
Sponsoring Organization/Resources
The GE2P2 Global Foundation’s Center for Informed Consent Integrity and its broader community of practice is leading this initiative, supported by the initiative advisory group above. The project benefits from strong support from the Alliance for Regenerative Medicine [ARM] and its Gene Editing Taskforce. Restricted grants resources are being sought from industry participants, patient advocacy groups, foundations, and other funders through the planned 2023 initiative period. Sangamo Therapeutics provided critical initial funding for this overall initiative via restricted grants, including support for a formative workshop held in collaboration with the New York Academy of Sciences in October 2019.
Initiative Timeline 2020-2025
[subject to pace of recruiting and access to IC content, etc.]
:: Recruit: industry, independent investigators, trial sites, patient organizations, other stakeholders
:: Collect: IC content and supporting materials, site interactions. Ongoing systematic lit reviews and engagements with scholars, research institutions and relevant organizations/collaborations.
:: Analyze: conduct comparative analysis of IC content and practices from above, report on findings
:: Draft: IC for Genomic Medicine Principles, refine through workshops with project community
:: Develop/test/refine: IC templates, toolkit and supporting content [training/interactives+]
:: Deploy: IC templates/toolkit/content in actual trials and clinical settings; assess/refine
Project Team [in formation; recruiting globally]
David R Curry, MS Co-PI
President, GE2P2 Global Foundation; Philadelphia, USA; Associate Faculty, Division of Medical Ethics, NYU School of Medicine, Philadelphia
Barbara Redman, PhD, MBE Co-PI
Senior Fellow; Director, Center for Informed Consent Integrity, GE2P2 Global Foundation, Washington, DC
Daima Bukini, BSc, MBE, MPH, PhD
Fellow- Bioethics, Muhimbili Univ of Health and Allied Sciences, Dar as Salaam
Paige Fitzsimmons, MA
Associate Director, Center for Informed Consent Integrity, GE2P2 Global, London
Jane S. Kang, MD, MS (Bioethics)
Associate Professor of Medicine, Columbia University Medical Center, New York City
Joanna Smolenski, PhD [Cand]
City University of New York, , New York City
Michael J. Young, MD, M.Phil
Center for Neurotechnology and Neurorecovery, Mass General | Harvard Med School, Boston
¡We are actively recruiting industry, individual investigators, trial sites, clinical settings, patient groups, all stakeholders to participate!
Contact: david.r.curry@ge2p2global.org +1.267.251.2305
open image
Project Resources

Bibliographic Materials [in development]: GE2P2 Global_CICI_ICGenMed_Bibliographic Materials_September 2020